- Blog
- Expert choice 11-5 serial number
- Cute writer pdf descargar gratis
- Arabic website for movies
- Fabfilter pro q 3 widen stereo bands
- Commercial pilot logbook
- Graphpad prism 8 user guide pdf
- Boot positivo unique s1991
- Load material cinema 4d
- Second law of thermodynamics statement
- Winebottler vs wineskin
- Advanced usb format tool
- Fxfactory mac
- Geek uninstaller pro serial
- Amaziograph for android download
- Muruga muruga om muruga muthamil iraiva vadivela lyrics
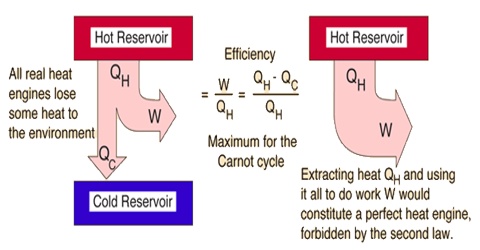
The force that makes the spontaneous reaction proceed in a certain direction is Entropy. FAQs on Entropy and Second Law of Thermodynamics.Kelvin-Planck Statement of the Second Law of Thermodynamics.Importance of Second Law of Thermodynamics.Entropy and Second Law of Thermodynamics.

Study Newton’s Second Law Of Thermodynamics Here However, before proceeding with the explanation for the second law of thermodynamics and what it represents, it is important to understand the Entropy of a system. Understanding the Entropy of a system and how it is related to the spontaneity of a system is what the second law of thermodynamics explains in detail. The spontaneity or the disturbance in the system, established by the term ‘entropy’, is taken into account in the second law of thermodynamics. The law could not explain the spontaneity or the feasibility of the reaction. The first law of thermodynamics establishes a relation between the heat changes and work done by a system, but it doesn’t discuss the direction of the flow of heat. Or “The total mass and energy of an isolated system remain unchanged.” See numerical problems based on this article.Entropy and Second Law of Thermodynamics: The first law of thermodynamics states that “Energy can neither be created nor destroyed it can only be transformed from one form to another” Hence, there is no change in its internal energy.īy the first law of thermodynamics we have – The working substance returns to its initial state after completing each cycle.The efficiency of a heat engine is defined as the ratio of net work done by the engine in one cycle to the amount of heat absorbed by the working substance from the source. The obtained work done ( W ) in each cycle is transferred to the environment by mechanical arrangement to get mechanical energy.The remaining amount of heat ( Q_2 ) is then released into the sink at temperature ( T_2 ).Then it converts a part of this heat energy into mechanical work ( W ).The working substance absorbs heat ( Q_1 ) from the source at temperature ( T_1 ).Freon or Ammonia is the working substance in a refrigerator or air conditioner.Ī heat engine works in four steps in every cycle of operation.Petrol or diesel is the working substance in an internal combustion engine.It can be a solid, a liquid or a gaseous substance.It is a substance which performs mechanical work when heat is supplied to it.It has infinite thermal capacity such that any amount of heat can be added to it and there will be no rise in temperature.It is a heat reservoir at a lower temperature ( T_2 ).

